SEE BETTER. TREAT BETTER.
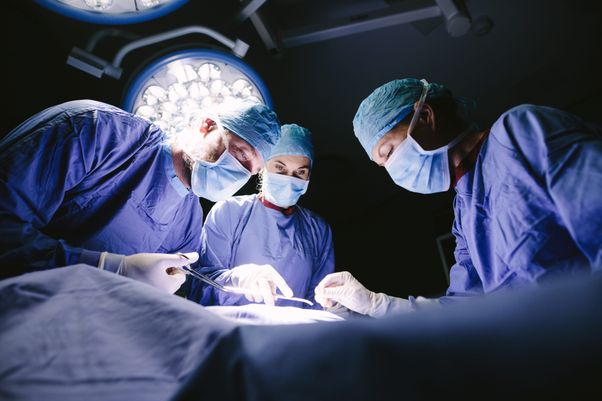
Permettre une visualisation et une détection plus précises pour renforcer la qualité et la fiabilité du diagnostic et du geste chirurgical grâce à nos colorants médicaux.
Depuis sa création, Provepharm accompagne l’évolution des usages grâce à son expertise en R&D, la performance et la haute qualité de ses colorants.
En développant des procédés de purification innovants, l’entreprise a créé et élevé les standards en matière de détection et de visualisation, favorisant l’émergence d’une nouvelle génération de colorants au service d’une médecine plus précise et personnalisée.
Colorants médicaux : des produits essentiels à haute technicité

Des solutions clés pour la visualisation
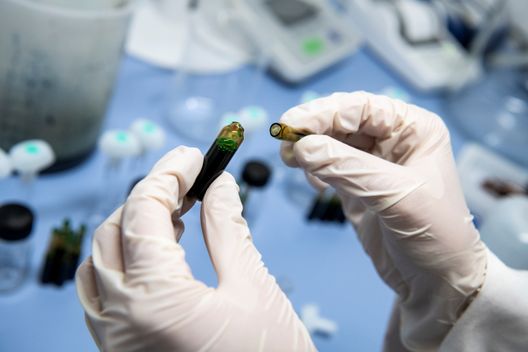
Utilisés en endoscopie et en chirurgie, les colorants médicaux sont des alliés précieux dans le repérage de certaines structures anatomiques ou de tissus pathologiques. Derrière ces produits essentiels, se cachent des procédés de synthèse complexes et un haut niveau d’exigence pharmaceutique.
Une expertise pointue en chimie organique et formulation
De la synthèse du principe actif à la formulation du produit fini, chaque étape nécessite une maîtrise fine de la chimie organique, de la galénique, et des normes de qualité les plus strictes.
Garantir disponibilité et performance
Notre expertise va au-delà de la R&D. Nous assurons :
- une production sécurisée et maîtrisée avec nos partenaires,
- une chaîne logistique fiable,
- la maitrise de l’approvisionnement auprès des hôpitaux en France et dans le reste du monde.
Un portefeuille unique au service des professionnels de santé
Nous proposons un portefeuille unique de colorants prêts à l’emploi, de haute qualité, développé pour répondre aux besoins cliniques, notamment en endoscopie et en chirurgie.
Découvrez notre gamme unique de colorants et produits hospitaliers
Découvrez notre gamme unique de colorants et produits hospitaliers
Médicaments et dispositifs médicaux de haute qualité, efficaces et prêts à l’emploi.
Provepharm, la référence mondiale des colorants médicaux au service de la chirurgie de précision
Recherche & Développement ciblée
au service d’une visualisation chirurgicale et détection améliorées
Maîtrise réglementaire et supply sécurisée
pour sécuriser l’accès marché et garantir une disponibilité fiable des produits
Accompagnement des professionnels de santé
pour répondre à leurs besoins et accompagner l’évolution des pratiques cliniques